[Exclusive] Health Ministry Cracks Down On Licence Rentals And Unsafe Prescription Practices
Doctors renting licences or selling prescription drugs without proper due diligence face fines and jail, MOH warns.

Follow us on Instagram, TikTok, and WhatsApp for the latest stories and breaking news.
Medical professionals caught renting out their licences or selling prescription drugs without proper due diligence face hefty fines and even imprisonment, the Health Ministry has warned
The warning follows a recent SAYS investigation, which found that some medical practitioners are dispensing prescription medications without proper checks.
The investigation also revealed that they had been leasing their Licensed Clinical Practitioner (LCP) licences to unqualified practitioners seeking to set up aesthetic clinics.
Read the full report here.
Meanwhile, the ministry's Pharmaceutical Services Programme told SAYS it takes all forms of unethical and unlawful conduct within the medical profession seriously, particularly practices that compromise patient safety and the integrity of healthcare services.
It said practices such as the "renting out" of Letters of Credentialing and Privileging (LCP) or the misuse of Annual Practising Certificates (APC) for unauthorised procurement of medicines are clear violations of professional and legal standards.
"The ministry remains steadfast in its commitment to uphold the highest standards of medical practice.
"Enforcement actions are taken decisively against any individuals or facility found to be in breach of the law, including investigations, show cause notices, compound fines and referrals to relevant authorities.
"Offenders may face disciplinary proceedings by the Malaysian Medical Council (MMC) and where applicable, prosecution under the Private Healthcare Facilities and Services Act 1998, the Poisons Act and the Sale of Drugs Act 1952.
"Penalties may include substantial fines and imprisonment," the ministry warned.

Image for illustration purposes only.
Image via NSTPNearly 300 unregistered and counterfeit diabetes drugs seized in raids
The ministry said its National Pharmaceutical Regulatory Agency (NPRA) has also received more than 100 local reports of side effects suspected to be linked to Ozempic and Rybelsus usage.
Over 80,000 reports have been recorded worldwide.
"Like other medicines in the same class, both products can cause side effects such as nausea, vomiting, diarrhoea, constipation and stomach discomfort, particularly at higher doses," it said.
The ministry said all pharmaceutical products are regulated under the Sale of Drugs Act 1952 and the Control of Drugs and Cosmetics Regulations 1984, requiring approval by the Drug Control Authority (DCA).
This, the ministry said, is to ensure that only products meeting strict quality standards, safety and efficacy reach the public.
"MOH remains committed to monitoring, investigating and taking enforcement actions against the distribution of unregistered and counterfeit medical products to safeguard public health and safety," it said.
The Pharmacy Enforcement Division (PED), it added, had also received 10 complaints between 2024 and 2025 concerning the online advertisement and sale of diabetes medications such as Ozempic and Rybelsus, all of which are under further investigation.
They made a disturbing discovery during enforcement raids – nearly 300 units of unregistered and counterfeit products containing semaglutide, the key ingredient in Ozempic and Rybelsus used to manage type 2 diabetes.
"Although there have been no seizures of Ozempic and Rybelsus to date, enforcement actions have resulted in the confiscation of 266 units of other unregistered or counterfeit products containing semaglutide, valued at RM19,420, through three raids conducted at a warehouse, a beauty spa centre, and a residential property.
"These actions were taken under the Poisons Act 1952 and the Control of Drugs and Cosmetics Regulations 1984," it said.
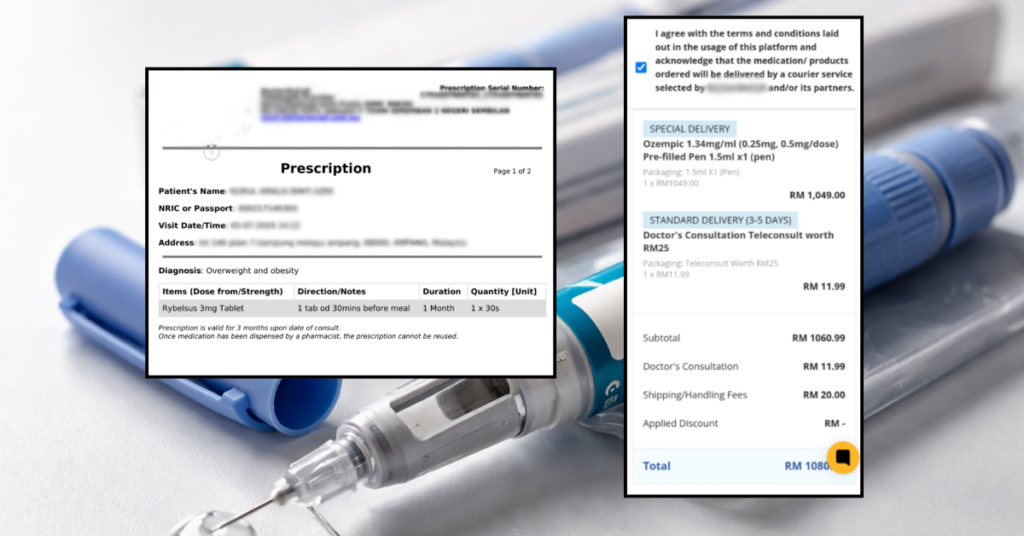
A telemedicine website allows easy access to prescribed medicine without thorough physical checks.
Image via SAYSMonitoring advertisements and telemedicine platforms
Even telemedicine platforms have not escaped the enforcement net, with 41 advertisements for Ozempic and 26 for Rybelsus screened between 2023 and September 19, 2025.
The PED stressed that it will continue to monitor, investigate and take firm action against any unlawful promotions under the Medicines (Advertisement and Sale) Act 1956.
"Where advertisements are found to contravene with the Medicines (Advertisement and Sale) act, action may be taken including investigation and prosecution in court, as well as the removal of non-compliant content from e-commerce platforms," it said.

Image for illustration purposes only.
Image via AI Generation/GeminiCrackdown on "renting out" of LCPs
The ministry acknowledged allegations of doctors "renting out" their LCPs to aesthetic clinics, stressing it treats such practices with the utmost seriousness.
"This practice is a serious breach of professional and legal standards and undermines the integrity of medical credentialing in Malaysia," the ministry said, adding that investigations are promptly launched whenever credible reports are received.
Both medical practitioners who "rent out" their LCPs and the facilities involved may face action under the Private Healthcare Facilities and Services Act 1998.
Doctors who are found guilty may also face disciplinary action by the Malaysian Medical Council (MMC).
The ministry said, in 2025 alone, 19 investigations have been conducted under Paragraph 31(1)(c) of the Private Healthcare Facilities and Services Act.
"Seven cases were given maximum compound fines of RM50,000 each, while others were issued show cause notices with the intention of revoking registration or licences under the act.
"It is also important to note that this offence carries a maximum penalty of two years imprisonment upon conviction," it said.

Every aesthetic practitioner is required to have an LCP to perform aesthetic procedures.
Image via SAYSMisuse of APCs by beauty centres
The ministry is also fully aware of attempts to exploit medical practitioners' APCs to obtain medicines.
The ministry said medical practitioners who allow their APCs to be misused will face the MMC for disciplinary action.
"Enforcement activities have also been undertaken by the Medical Practice Division, either through surveillance or upon receiving complaints, under Section 4 of the Private Healthcare Facilities and Services Act 1998.
"In 2025 alone, 58 cases have been investigated under this provision, with 25 investigation papers formally opened for further action," it said.
Those found guilty under the section could face a fine of up to RM300,000, imprisonment for up to six years, or both.
The highest penalty imposed by the court to date was a fine of RM100,000 or two years' imprisonment.
The ministry added that between 2020 and 2025, authorities conducted 153 raids on beauty and spa premises, seizing 19,905 units of products containing controlled substances valued at RM957,907.
While, 2022 recorded the highest number of confiscated products, 2025 saw a marked increase in the overall value of seizures, reflecting intensified enforcement.
Safeguarding patient safety
As part of ongoing patient safety initiatives, NPRA has introduced e-labelling, enabling consumers and healthcare professionals to access up-to-date information via QR codes on product packaging.
The ministry said a directive has also been issued to update package inserts of GLP-1 receptor agonist products, including semaglutide, to include warnings about risks of aspiration and aspiration pneumonia during general anaesthesia or deep sedation.
The ministry urged the public to seek aesthetic and medical services only from licensed healthcare facilities with properly credentialed practitioners.
To verify private healthcare facilities and LCP holders, the public can refer to https://hq.moh.gov.my/medicalprac/statistik/ for private healthcare facilities status or https://www.moh.gov.my/index.php/pages/view/2118 for Letter of Credentialing and Privileging (LCP) holder status.
Complaints or reports of irregular or unsafe practices can be lodged with the Private Medical Practice Control Section (CKAPS) via email at [email protected] or through their portal at https://myckaps.spab.gov.my/eApps/system/index.do.


